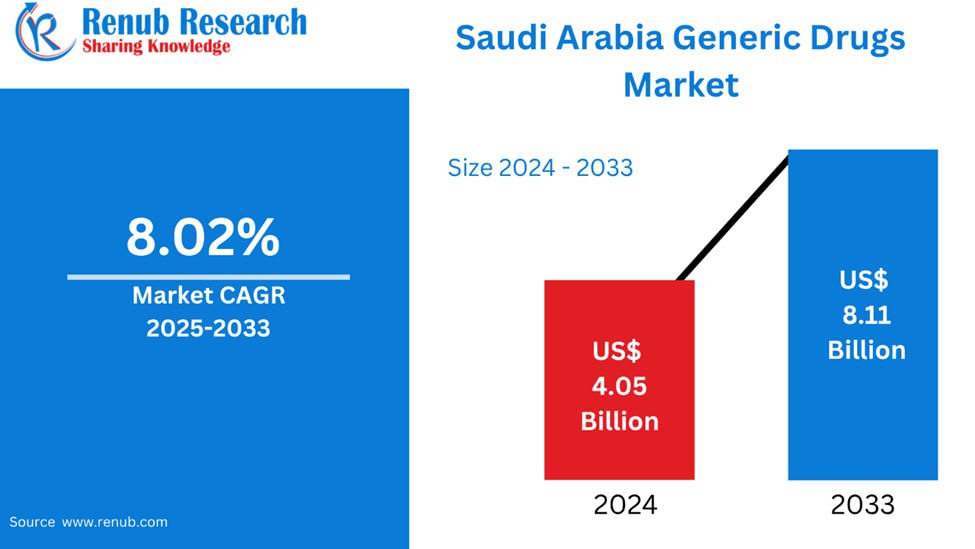
Saudi Arabia Generic Drugs Market is anticipated to grow to US$ 8.11 billion by 2033 from US$ 4.05 billion in 2024, growing at a CAGR of 8.02% during 2025 to 2033. This growth is attributed to increased healthcare needs, government efforts to curb pharmaceutical expenditure, and mounting awareness for cost-effective alternatives. The market is also boosted by growing local manufacturing and government support for generics.
Saudi Arabia Generic Drugs Market Overviews
Generic drugs are bioequivalent medications that have the same active ingredients, dosage strength, dosage form, and route of administration as their brand-name equivalents but at lower prices. They are licensed by regulatory bodies only after proving their bioequivalence to branded medicines, guaranteeing they have the same therapeutic effects. Generic drugs lower the cost of healthcare substantially and promote access to necessary medication.
In Saudi Arabia, the demand for generic medications is increasing at a very high pace. It is fueled by attempts by the government to lower reliance on imported branded medicines and in trying to slash healthcare spending. The Saudi Food and Drug Authority (SFDA) has made the process of generic approvals easier, prompting local and foreign manufacturers to increase their generic offerings. Additionally, public health awareness campaigns and a focus on preventive care have supported the acceptance of generic medicines among consumers and healthcare professionals. With a growing population and rising incidence of chronic diseases, the demand for cost-effective generic drugs is expected to expand further across the Kingdom.
Growth Drivers in the Saudi Arabia Generic Drugs Market
Government Support and Cost Containment Initiatives
The Saudi government has focused on making healthcare more affordable through greater generic drug promotion to lower the country’s healthcare expenditure. More rapid regulatory approval and increased local manufacturing capacity have been promoted through initiatives spearheaded by the Saudi Food and Drug Authority (SFDA). Cost-effective treatment is now more favored in national health policy, with generics more extensively used in public hospitals and among insurance companies. These initiatives have considerably fueled the country’s generic drug market and brought in foreign pharmaceutical investment into the Kingdom of Saudi Arabia. The Saudi Food and Drug Authority (“SFDA”) released a draft initiative, the “Procedure to deal with patents when registering generic products in SFDA”. The Initiative has been made available on the Saudi Public Consultation Platform website for consultancy and the SFDA has invited the public and pharmaceutical firms in June 2022.
Increasing Incidence of Chronic Diseases
With increasing numbers of chronic diseases like diabetes, cardiovascular disease, and respiratory diseases, Saudi Arabia is in need of long-term affordable medication to a greater degree. Generic drugs are a viable solution to costly branded drugs for the management of chronic diseases. With the population growing older and living lifestyles that are inactive, the demand for efficient, cost-effective medication is encouraging patients and doctors to use generics, compelling steady growth in demand in the industry. April 2025, Among Middle Eastern countries, Saudi Arabia has one of the highest rates of chronic diseases (CDs) at 32.15%. This creates huge healthcare challenges to the population. The most common causes of morbidity are diabetes mellitus (DM), hypertension, and obesity. The rise in the incidence of these chronic diseases adversely impacts the quality of life of patients and increases total healthcare expenditure.
Increase in Local Production Capability
Saudi Arabia has made significant investments in developing pharmaceutical production facilities under Vision 2030 to increase local production and minimize import reliance. With new plants set up with partnerships and technology transfers, generic drug production is growing very fast. This local presence allows for quicker availability, reduced prices, and more control over quality. The domestic boom in production is making the Kingdom a competitive force in the regional generic pharma market. In January 2024, Saudi Arabia announced a national biotech plan to become a world-class biotech hub in 16 years. The program is part of a bigger effort known as Vision 2030, which aims to position the country as a biotech leader in the Middle East and North Africa by 2030.